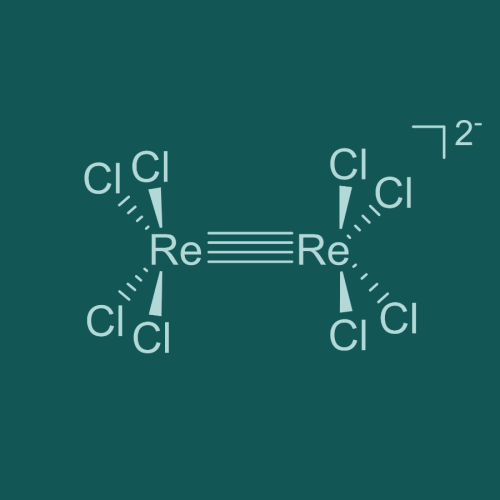
Octachlorodirhenate
Other names: dirhenium(III) octachloride, octachloridodirhenate(III)
[Re2Cl8]2-
49/50  rare
rare
Typically found as the potassium salt K2[Re2Cl8], this simple complex features a quadruple bond between the two rhenium centers, courtesy of a δ bond between the two 4dxy orbitals in the rhenium centers. Although this species represents a fairly elegant example of metal-metal multiple bonding, it is far from the only example of the type; metal-metal bond orders as high as 5 (quintuple bonding) have been observed experimentally and sextuple bonding is theorized to exist in gas-phase dimolybdenum (Mo2) and ditungsten (W2) at temperatures above 4500 °C. Even higher-order bonding implicating f orbitals (φ bonding) has long been theorized, though despite decades of theorizing and computational modeling no diatomic bond orders above 6 are definitively known to exist.