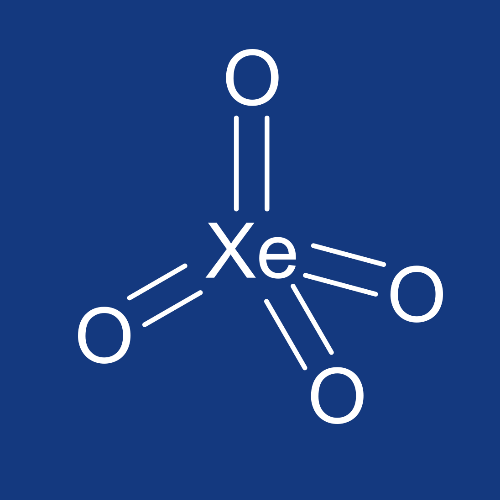
Xenon tetroxide
Other names: xenon (VIII) oxide, perxenic anhydride
XeO4
17/50  common
common
Although the defining chemical feature of the noble gases is their unreactivity, the heavier members of this group (particularly krypton and xenon) are indeed capable of forming simple compounds upon reaction with sufficiently oxidizing species. This is because, although all noble gases possess a full octet in their neutral monatomic state, the valence electrons for heavier noble gases are sufficiently shielded and distanced from the nucleus that they may be abstracted by electron-hungry elements such as fluorine and oxygen. Xenon tetroxide is synthesized by the dehydration of perxenic acid, H4XeO6. The resultant XeO4 is an extremely unhappy solid at temperatures below -36 °C, and decomposes violently into gaseous xenon and oxygen at temperatures above that point.